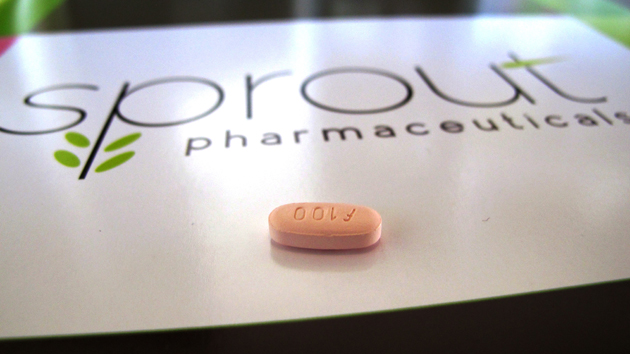
Flibanserin, a drug to treat low sexual desire in women.Allen Breed/AP
A contentious effort to introduce a sex-drive drug for women may be nearing a climax. After rejecting “the pink Viagra” (AKA flibanserin) twice in the past five years, a Federal Drug Administration advisory committee voted today that the the pill should be approved if measures are taken to reduce its potential side effects, such as low blood pressure and fainting.
The nonhormonal drug, developed by Sprout Pharmaceutical, is meant to be taken nightly. The company says it works by altering chemicals in the brain to increase a woman’s sexual desire (unlike Viagra, which increases blood flow to certain parts of the body). Women who took the drug in trials reported no more than one additional “sexually satisfying event” per month than women who received a placebo.
Today’s vote comes 17 years after the FDA approved Viagra, the incredibly profitable erectile dysfunction treatment. Since then, more than two dozen prescription drugs have been approved to assist with male sexual performance, but none for women.
Even the Score, a group backed by the pharmaceutical companies that developed flibanserin, says that women also suffer from sexual dysfunction and that the unwillingness to develop and approve drugs to treat it is a matter of bias. They called today’s vote a victory for gender equality. “Women with Hypoactive Sexual Desire Disorder deserve the safety and peace of mind that comes with access to FDA-approved medical treatment options, and today we write a new chapter in the fight for equity in sexual health,” Even the Score said in a statement.
But others believe that potentially profitable drugs for a dubious disorder are being pushed with little proven effectiveness. “I don’t think there is anything sexist about denying approval for drugs that don’t have an adequate risk-to-benefit ratio,” Thea Cacchioni, a professor of women’s studies at the University of Victoria in British Columbia, told the New York Times. According to Forbes, $50 million in private investment rest on flibanserin’s approval.
The FDA is expected to come to a final decision on the drug in August.